|
10/20/2023 0 Comments Molar entropyChemical Engineering Research Information Center. ^ a b c "Binary Vapor-Liquid Equilibrium Data" (Queriable database).Archived from the original (Queriable database) on. Advanced Industrial Science and Technology. ^ a b c "Spectral Database for Organic Compounds".^ Lange's Handbook of Chemistry 10th ed, pp. 1522–1524.^ a b CRC Handbook of Chemistry and Physics 44th ed.^ a b Lange's Handbook of Chemistry 10th ed, pp. 1525–1528."Molar Heat Capacity (cP) of Methanol from Dortmund Data Bank". ^ "Vapor Pressure of Methanol from Dortmund Data Bank".^ Lange's Handbook of Chemistry, 10th ed.^ NMR-002: Sample Devices and Magnetic Susceptibility.When temperature b is 4 ☌, density of water is 0.999972 g/mL.ĭistillation data Vapor–liquid equilibrium The annotation, d a☌/ b☌, indicates density of solution at temperature a divided by density of pure water at temperature b known as specific gravity. and CRC Handbook of Chemistry and Physics 44th ed. Properties of aqueous methanol solutions ĭata obtained from Lange's Handbook of Chemistry, 10th ed. Note that the form of this formula as given is a fit to the Clausius–Clapeyron equation, which is a good theoretical starting point for calculating saturation vapor pressures: Here is a similar formula from the 67th edition of the CRC handbook. Uses Antoine's equation: P m m H g = 10 7.87863 − 1473.11 230.0 + T from Lange's Handbook of Chemistry 10th ed. Table data obtained from CRC Handbook of Chemistry and Physics 44th ed. 
Thermodynamic properties Phase behaviorħ0.8–90.5 J/(mol K) (at −97.6 to 64.7 ☌) Baker and Loba Chemie Structure and properties Structure and properties Uses formula: for T 0 to 36 C for T 36 to 170 C Formula from Lange's Handbook of Chemistry, 10th ed. It is highly recommended that you seek the Safety Datasheet ( SDS) for this chemical from a reliable source such as SIRI, and follow its directions. Thermodynamic properties Vapor pressure of liquid Table data obtained from CRC Handbook of Chemistry and Physics 44th ed. 
The handling of this chemical may incur notable safety precautions. (eds.This page provides supplementary chemical data on methanol. It is highly recommend that you seek the Safety Data Sheet ( SDS) for this chemical from a reliable source and follow its directions. Heat capacity of anhydrous liquid ammonia. Heat capacity of liquid and vapor Heat capacity, c p, of anhydrous ammonia gas. The fifth column is the heat of vaporization needed to convert one gram of liquid to vapor.įreezing curve of ammonia-water system. The fourth column is the density of the vapor. The third column is the density of the liquid phase. The second column is vapor pressure in k Pa. 
The table above gives properties of the vapor–liquid equilibrium of anhydrous ammonia at various temperatures. Where P is pressure in k Pa, and T is temperature in kelvins Ī = 6.67956, B = 1002.711, C = 25.215 for T = 190 K through 333 K. (18.10).The molar enthalpy and entropy of vaporisation and the boiling temperature of some simple liquids are presented in Table 18.2 Some liquids deviate sharply from the rule. Vapor-pressure formula for ammonia: log 10 P = A – B / ( T − C), S vap.m is equal to ratio between the molar enthalpy of vaporisation and the boiling temperature, which leads to Eq. Log 10 of anydrous ammonia vapor pressure. Otherwise temperature is equilibrium of vapor over liquid. The differences in heat capacities on melting and on vaporization are 37.3 J K1mol1 and 41.9 J K1 mol1, respectively. 19, we obtain the following expression for the molar. 3.1 Calculate the difference in molar entropy (a) between liquid water and ice at 5C, (b) between liquid water and its vapour at 95C and 1.00 atm. (20) S S v + S t + S r Substituting the vibrational partition function given in Eq. The (s) notation indicates equilibrium temperature of vapor over solid. The entropy is given by : (19) S k B l n Q + k B T ( l n Q V) T The molar entropy is a combination of three contributions, including vibrational, translational and rotational entropies. All of them have tetrahedron structure and the four corners are identical so there is only one way to organize CHF3. The trend here is based on molecular complexity. 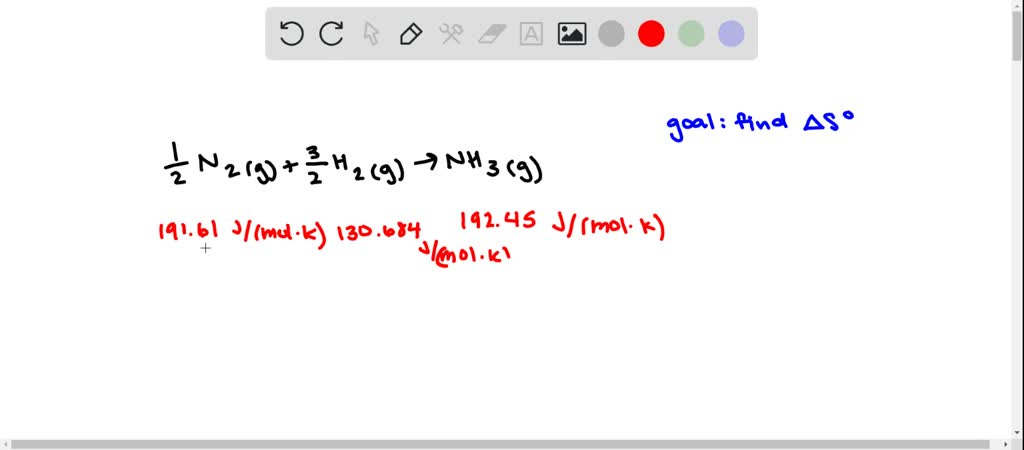
Table data (above) obtained from CRC Handbook of Chemistry and Physics 44th ed. Re: Arrange in order of increasing standard molar entropy. Thermodynamic properties Phase diagram and crystalline states of ammonia Structure and properties Molecular structure This page provides supplementary chemical data on ammonia.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
